-
半导体泵浦碱金属蒸汽激光器(DPAL)由半导体激光泵浦钾(K)、铷(Rb)和铯(Cs)等碱金属蒸汽以实现近红外激光输出[1]。它具有量子效率高、吸收截面大、气体介质折射率扰动小、热管理便捷和可定标放大等优点,与传统固体激光器相比,DPAL可通过碱金属蒸汽的循环流动来降低热效应,散热性能好,具有高功率、高效率、高光束质量激光输出的优势。
DPAL作为三能级激光系统,需要加入缓冲气体来加速碱金属原子精细结构能级之间的快速弛豫,因此要求缓冲气体分子的转动能隙接近碱金属原子精细结构能级之间能量差。对于K和Rb蒸汽激光器,使用惰性气体(He、Ar等)和烷烃气体(CH4、C2H6等)均能满足上述要求,但是考虑到高温下烷烃气体会与碱金属蒸汽发生反应,因此目前实验中大多使用惰性气体(主要为He)作为缓冲气体[2-4];对于Cs蒸汽激光器(Cs-DPAL),烷烃气体的转动能隙与Cs原子精细结构能级之间能量差较为接近,而惰性气体的转动能隙则相差较多,只使用惰性气体作为缓冲气体无法实现Cs原子精细结构能级之间的快速弛豫,因此必须使用烷烃气体作为缓冲气体[3],而烷烃气体和惰性气体均可以对Cs原子的吸收谱进行展宽,因此均可使用纯烷烃气体或者烷烃气体和惰性气体的混合物作为Cs-DPAL的缓冲气体。
2012年,俄罗斯联邦核子中心使用He和CH4作为缓冲气体,首次实现了千瓦量级循环流动Cs蒸汽激光输出,光光转换效率高达48%[5]。2016年,美国空军学院Knize等人使用烷烃气体和烷烃与惰性气体的混合物进行了对比实验,发现烷烃气体压强大于200 torr (1 torr≈133.322 Pa)且总缓冲气体压强大于300 torr时,使用纯CH4、纯C2H6或C2H6和He的混合物作为缓冲气体对循环流动Cs-DPAL输出影响差异不大[6]。2017年,日本东海大学Yamamoto等人分别使用He和Ar与烷烃气体混合作为缓冲气体进行循环流动Cs-DPAL实验,发现在泵浦功率为13 W的条件下,气体流速小于0.5 m/s时,相比于Ar,He作为缓冲气体,DPAL输出功率明显更高;在气体流速大于6 m/s时,二者对输出的贡献无明显差异[7]。2018年,以色列古里安大学Yacoby等人使用He和CH4作为缓冲气体进行了连续波循环流动Cs-DPAL实验,在泵浦功率为65 W时获得了24 W的最大连续激光输出,斜效率达到48%[8]。
目前,上述理论和实验研究均未考虑缓冲气体对高功率循环流动Cs-DPAL气体温度三维分布的影响,蒸汽池内的气体温度分布是影响激光器性能的关键指标,过高的气体温度和温度梯度会导致严重的热效应。因此,文中将在理论上分析缓冲气体成分和气体压强对高功率循环流动Cs-DPAL气体温度三维分布的影响,进而分析缓冲气体对激光输出性能的影响。考虑到激光能量分布也会对气体温度分布产生影响,文中首先在速率方程、流体力学方程与热平衡方程基础上,首次将光束传播方程引入循环流动Cs-DPAL理论模型,分析高功率泵浦情况下,纯烷烃气体(CH4和C2H6)和烷烃与惰性气体(He和Ar)的混合气对循环流动Cs-DPAL蒸汽池内气体温度三维分布和输出特性的影响,并获得了缓冲气体的最佳成分配比和压强,为高功率循环流动Cs-DPAL实验中缓冲气体的选取提供理论指导。
-
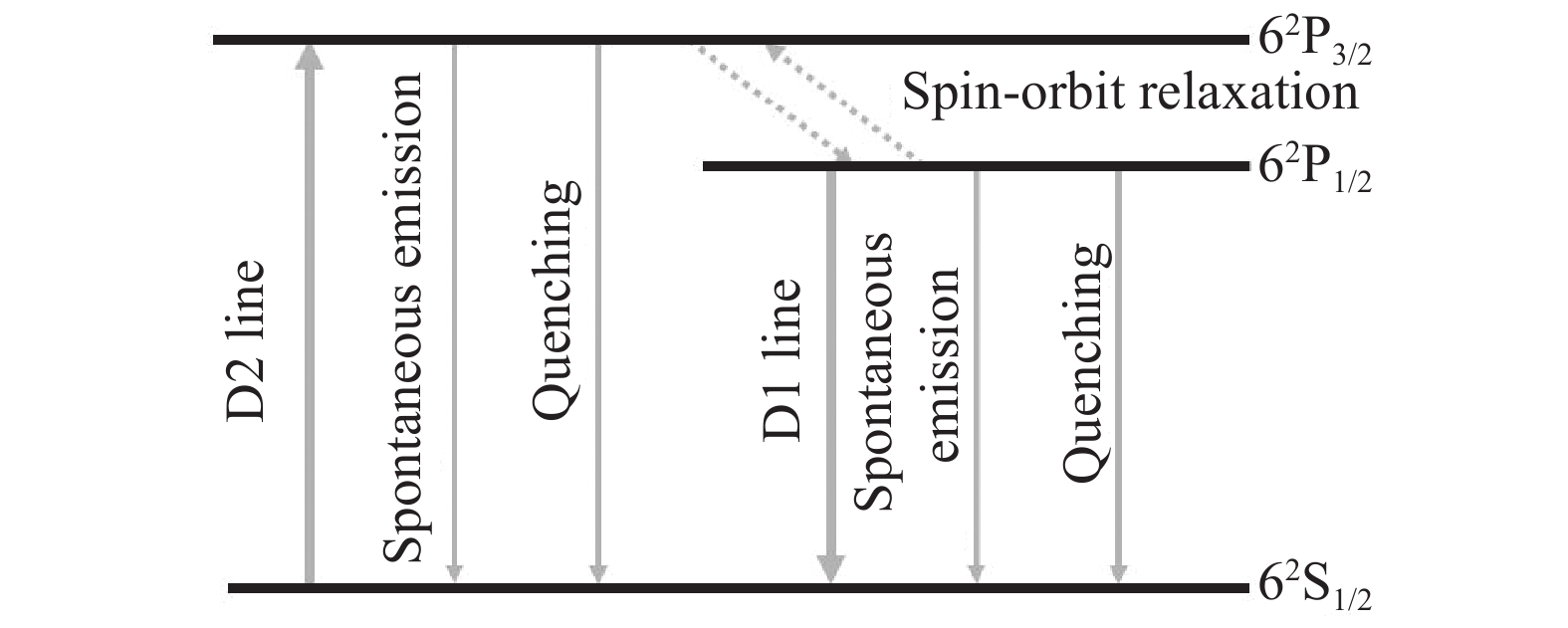
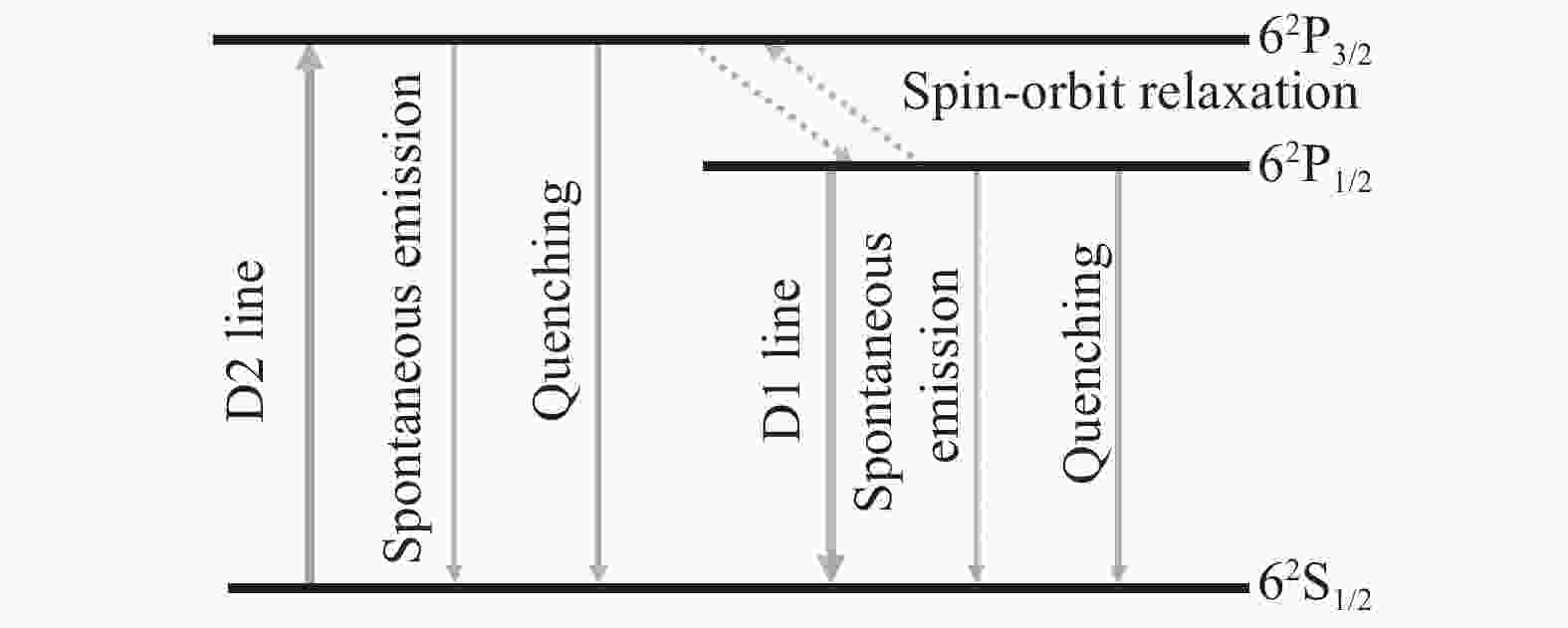
图1为Cs原子的能级跃迁图,其中,D2线表示Cs原子吸收泵浦光子后由基态62S1/2跃迁至泵浦上能级62P3/2的过程;Cs原子再通过无辐射弛豫跃迁至激光上能级62P1/2,该过程即为精细结构能级间的弛豫过程;D1线表示Cs原子从激光上能级62P1/2回到基态能级62S1/2并辐射出激光光子的过程。其他的动力学过程还包括碱金属原子的自发辐射和电子态淬灭过程,相较于自发辐射,电子态淬灭对激光系统的影响可忽略不计[9]。
忽略蒸汽池内碱金属原子与烷烃气体发生反应和向高能级跃迁,DPAL的速率方程可以用以下方程表示:
$$ \begin{gathered} \frac{{{\text{d}}{{{n}}_3}}}{{{\text{d}}t}} = \frac{{{\sigma _p}\left( {{n_1} - \dfrac{1}{2}{n_3}} \right){I_p}}}{{h{\nu _p}}} - {\gamma _{32}}\left( {{n_3} - 2{n_2}\exp \left( { - \frac{{\Delta E}}{{{k_B}T}}} \right)} \right) - \frac{{{n_3}}}{{{\tau _3}}} \\ \frac{{d{n_1}}}{{{\text{d}}t}} = - \frac{{{\sigma _p}\left( {{n_1} - \dfrac{1}{2}{n_3}} \right){I_p}}}{{h{\nu _p}}} + \frac{{{\sigma _l}\left( {{n_2} - {n_1}} \right){I_l}}}{{h{\nu _l}}} + \frac{{{n_3}}}{{{\tau _3}}} + \frac{{{n_2}}}{{{\tau _2}}} \\ {n_0}\left( T \right) = {n_1}\left( T \right) + {n_2}\left( T \right) + {n_3}\left( T \right) \\[-6pt] \end{gathered} $$ (1) 式中:n1、n2、n3分别表示蒸汽池内温度为T时基态能级62S1/2、激光上能级62P1/2、泵浦上能级62P3/2的粒子数浓度;n0为蒸汽池内温度为T时对应的总粒子数浓度,假设腔壁处温度为Tw,总粒子数浓度为nw ,则有:
$$ {n_0} = {n_w}\frac{{{T_w}}}{T} $$ (2) $I_p $ 和$I_l $ 分别为泵浦和激光光强;$ v_p $ 和$v_l $ 分别为泵浦和激光频率;$ {\sigma _p}$ 和${\sigma _l} $ 分别表示泵浦吸收截面和激光发射截面;$\gamma _{32}$ 为精细结构62P3/2~62P1/2的弛豫速率;$\Delta E$ 为精细结构62P3/2和62P1/2之间能级差;$ k_B $ 为玻耳兹曼常数;T为蒸汽池内碱金属蒸汽温度。在稳态情况下,公式(1)微分项均为零,给定一个泵浦和激光光强即可求得各能级粒子数浓度。求得粒子数浓度之后,可通过公式(3)求得腔内泵浦吸收系数和激光增益系数:
$$ \begin{gathered} {g_p}\left( {x,y,z} \right) = {\sigma _p}\left[ {{n_1}\left( {x,y,z} \right) - \frac{1}{2}{n_3}\left( {x,y,z} \right)} \right] \\ {g_l}\left( {x,y,z} \right) = {\sigma _l}\left[ {{n_2}\left( {x,y,z} \right) - {n_1}\left( {x,y,z} \right)} \right] \\ \end{gathered} $$ (3) 通过光束传播方程来求解蒸汽池中被吸收的泵浦光振幅
$A_p $ [10]:$$ \begin{split} 2i{k_0}\frac{{\partial {A_p}\left( {x,y,z} \right)}}{{\partial z}} =& \frac{{{\partial ^2}{A_p}\left( {x,y,z} \right)}}{{\partial {x^2}}} + \frac{{{\partial ^2}{A_p}\left( {x,y,z} \right)}}{{\partial {y^2}}}- \\ & ik_0^2\frac{{\text{1}}}{{{k_0}}}{g_p}\left( {x,y,z} \right){A_p}\left( {x,y,z} \right)\\[-8pt] \end{split} $$ (4) 蒸汽池中传播的激光振幅
$A_l $ 可表示为:$$ \begin{split} 2i{k_0}\frac{{\partial {A_l}\left( {x,y,z} \right)}}{{\partial z}} =& \frac{{{\partial ^2}{A_l}\left( {x,y,z} \right)}}{{\partial {x^2}}} + \frac{{{\partial ^2}{A_l}\left( {x,y,z} \right)}}{{\partial {y^2}}} + \\ & ik_0^2\frac{1}{{{k_0}}}{g_l}\left( {x,y,z} \right){A_l}\left( {x,y,z} \right) \\[-8pt] \end{split} $$ (5) 自由空间光路中传播的激光振幅可表示为:
$$ 2i{k_0}\frac{{\partial {A_l}\left( {x,y,z} \right)}}{{\partial z}} = \frac{{{\partial ^2}{A_l}\left( {x,y,z} \right)}}{{\partial {x^2}}} + \frac{{{\partial ^2}{A_l}\left( {x,y,z} \right)}}{{\partial {y^2}}} $$ (6) 式中:k0为真空中光的波数;x和y为振荡激光横截面坐标;z为激光振荡方向坐标,通过求解泵浦和激光振幅,即可得到输出激光光强。
$$ \begin{split} {P_{waste}} =& \iiint\limits_{{V_l}} {\left\{ {{\gamma _{32}}} \right.\left[ {{n_3} - 2{n_2}\exp \left( { - {{\Delta E} \mathord{\left/ {\vphantom {{\Delta E} {{k_B}T}}} \right. } {{k_B}T}}} \right)} \right]\Delta E} + \\ & h\nu \left( {{Q_{21}} + {Q_{31}}} \right) + \left. {{R_{rec}}{E_i}} \right\}{\rm d}x{\rm d}y{\rm d}z \\ \end{split} $$ (7) 式中:
$V_l $ 为激光的模体积;Rrec为离子复合速率;Ei为电离能。公式(7)右侧第1项对应62P3/2能级弛豫至62P1/2能级所产生的废热,第2项对应62P1/2能级和62P3/2能级无辐射跃迁至基态所产生的废热,第3项对应离子复合过程所产生的废热。由于62D5/2,3/2能级和82S1/2能级至基态的无辐射跃迁所产生的废热极小,因此计算中忽略该部分对废热的影响。工作气体流动时的散热量可表示为[12]:
$$ {R_{heat}} = \frac{{{\text{2}}{\omega _l}Lu{N_w}}}{{Na}}\int\limits_{{T_w}}^T {{C_p}\left( {{T'}} \right)} {\rm d}{T'} + 2\pi {\omega _l}k\left( T \right){N_u}\left( {T - {T_w}} \right) $$ (8) 式中:Nw为管壁附近碱金属蒸汽与缓冲气体的总粒子数密度;Nu为努塞尔数;
$C_p(T) $ 和$k(T) $ 分别为总缓冲气体的摩尔热容和热导率。当激光器系统达到热平衡状态时,蒸汽池内的产热量等于散热量:
$$ {P_{waste}} = {R_{heat}} $$ (9) 将公式(7)和(8)代入上式,即可求得稳态下碱金属蒸汽池内的温度分布。
假设碱金属蒸汽池中的初始激光光强为0,碱金属蒸汽池内气体初始温度分布均匀,泵浦光初始振幅
$A_p $ 和蒸汽池内气体初始温度已知。对于本循环流动Cs-DPAL理论模型,具体数值计算过程如下:(1) 通过求解速率方程得到碱金属原子粒子数浓度的三维分布,再求解泵浦和激光增益系数,通过光束传播方程得到泵浦光与激光的三维分布。
(2) 通过流体力学与热平衡求得温度的三维分布。
(3) 将求得的这三者的值代替给定的初值,不断重复上述计算过程。
(4) 计算至三者均趋于稳定,得到泵浦光振幅、激光振幅和腔内气体温度的三维分布。
-
由于横向流动时气体的散热效果较好,输出功率较高[13],因此对横向流动Cs-DPAL进行仿真分析。
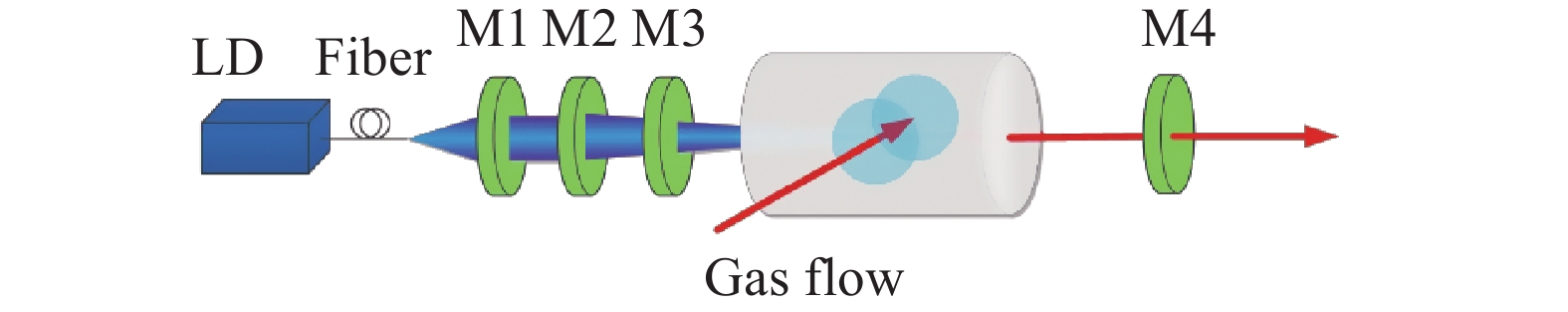
图2为端面泵浦横向流动Cs-DPAL示意图,泵浦源为波长852 nm的光纤耦合LD,光纤数值孔径为0.22,纤芯半径为200 μm。泵浦光从光纤输出后通过聚焦镜M1进行准直,再通过M2聚焦进入Cs蒸汽池端面,M1和M2均镀852 nm波段高透膜。Cs蒸汽池内含有金属Cs和缓冲气体,循环时Cs蒸汽与缓冲气体混合在蒸汽池内垂直于光轴方向流动,蒸汽池长度为2 cm,蒸汽池两端窗口镜半径均为1 cm,均对852 nm和895 nm波段高透。温控炉控制Cs蒸汽池侧壁的工作温度为373 K。M3为平面二色镜,对852 nm波段高透,895 nm波段高反,M4为平面输出镜,对895 nm波段反射率50%。具体仿真参数如表1所示。
表 1 循环流动Cs-DPAL仿真参数
Table 1. Parameters of gas flowing diode pumped cesium laser
Parameters Value Parameters Value Central wavelength of pump light/nm 852.3 Waist radius of pump light/mm 1 Pump light linewidth/GHz 70 Pump light transmittance of M1 and M2 100% Pump light transmittance of M3 99% Laser reflectivity of M4 50% Distance between waist of pump light and central of vapor cell/mm 0 Sidewall temperature of vapor cell/℃ 100 Distance between vapor cell and M3/mm 50 Distance between vapor cell and M4/mm 50 Gain length of vapor cell/mm 20 Velocity of flowing gas/m·s−1 20 文中选择了两种烷烃气体(CH4和C2H6)和两种惰性气体(He和Ar)作为缓冲气体进行仿真研究。表2给出了在压强为1000 torr、温度为450 K时上述缓冲气体的摩尔热容、热导率和粘滞系数[14-18]。
表 2 缓冲气体的摩尔热容、热导率和粘滞系数
Table 2. Molar heat capacity, thermal conductivity and viscosity of buffer gases
Buffer gases Molar heat capacity/J·mol−1·K−1 Thermal conductivity/ W·m−1·K−1 Viscosity/ Pa·s−1 Ethane 72.027 4.464×10−2 1.353×10−5 Methane 43.550 5.906×10−2 1.566×10−5 Helium 20.785 2.066×10−1 2.636×10−5 Argon 20.810 2.451×10−2 3.138×10−5 -
首先使用纯CH4和C2H6作为缓冲气体对Cs-DPAL气体温度分布和激光输出特性进行研究,图3给出了1 kW泵浦情况下,使用纯CH4和纯C2H6作为缓冲气体时,蒸汽池内温度三维分布与缓冲气体压强之间的关系,气体流速为20 m/s。蒸汽池左端为泵浦光输入窗口,右端为激光输出窗口。可见,由于激光在蒸汽池中心振荡,因此蒸汽池内中心处气体温度最高,且温度从中心至池壁不断降低。同时,蒸汽池左侧中心处气体温度最高,这是由于泵浦光从左侧注入,Cs蒸汽吸收的泵浦光功率最高。在CH4和C2H6均为400 torr时,蒸汽池内最高温度分别为左侧输入窗口中心处的655.3 K和694.4 K。当压强升至700 torr和1000 torr时,CH4对应的蒸汽池内最高温度分别为596.4 K和540.5 K,C2H6对应的蒸汽池内最高温度分别为557.8 K和506.1 K。对比可知,相同压强下,C2H6对应的气体温度要低于CH4,这是因为C2H6作用于Cs原子的弛豫截面大于CH4,导致相同情况下精细结构弛豫速率更高,泵浦光转换效率更高,热量沉积更少。此外,随着缓冲气体压强的增加,腔内温度随之下降。这是因为在压强较低时,精细结构自旋轨道弛豫速率较小,泵浦吸收系数和激光增益系数均较低,大量泵浦光无法转换为激光输出而转化为热量沉积在蒸汽池内,导致腔内温度较高;随着压强的增加,泵浦光转换效率不断提升,蒸汽池内废热量减少,温度随之降低。
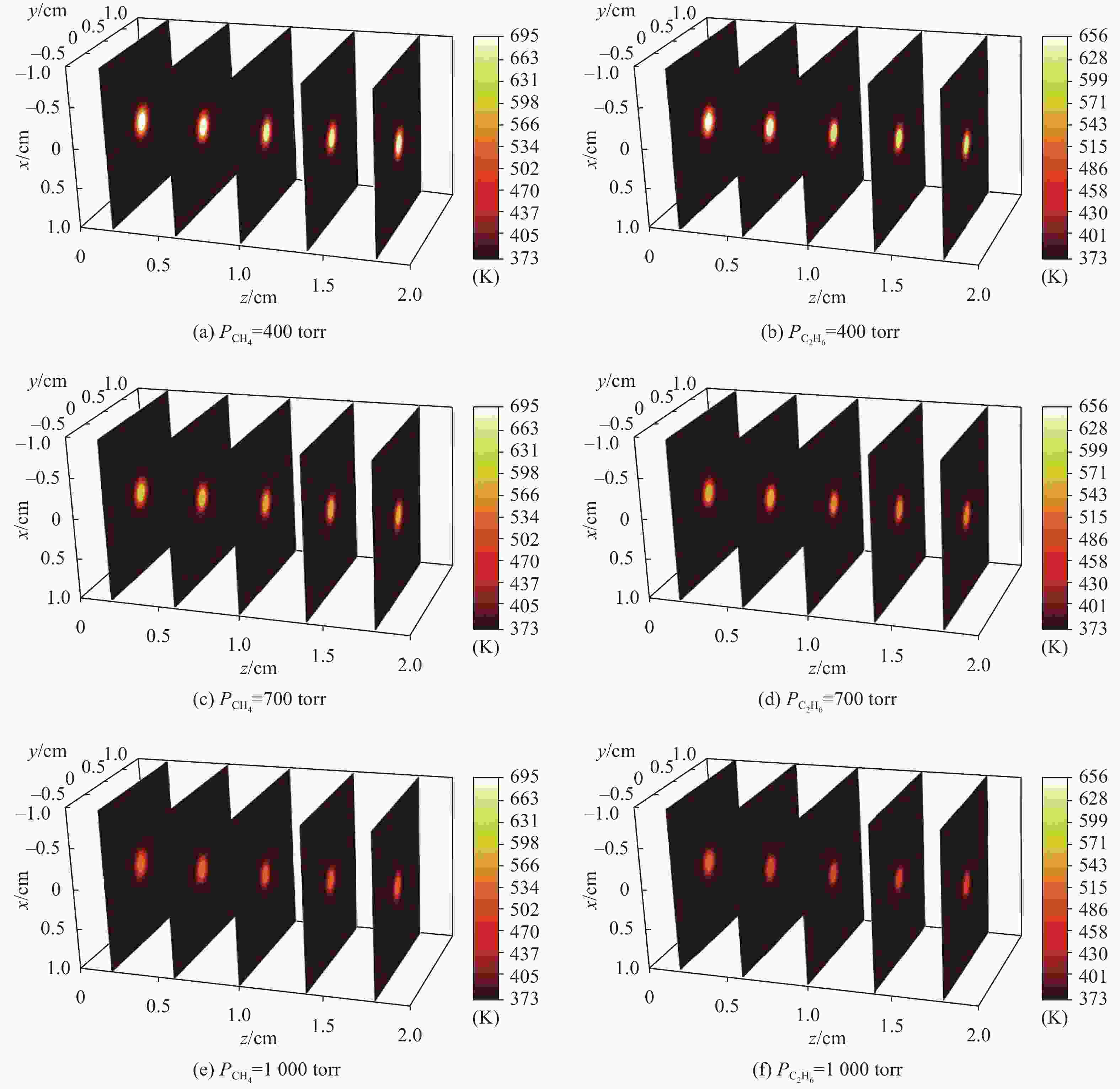
图 3 蒸汽池内温度分布与缓冲气体压强之间的关系。(a)、(c)、(e)对应CH4压强分别为400 torr、700 torr和1000 torr;(b)、(d)、(f)对应C2H6压强分别为400 torr、700 torr和1000 torr
Figure 3. Relationship between temperature distribution and buffer gas pressure in vapor cell. (a), (c) and (e) correspond to 400 torr, 700 torr and 1000 torr methane pressures, respectively; (b), (d) and (f) correspond to 400 torr, 700 torr and 1000 torr ethane pressures, respectively
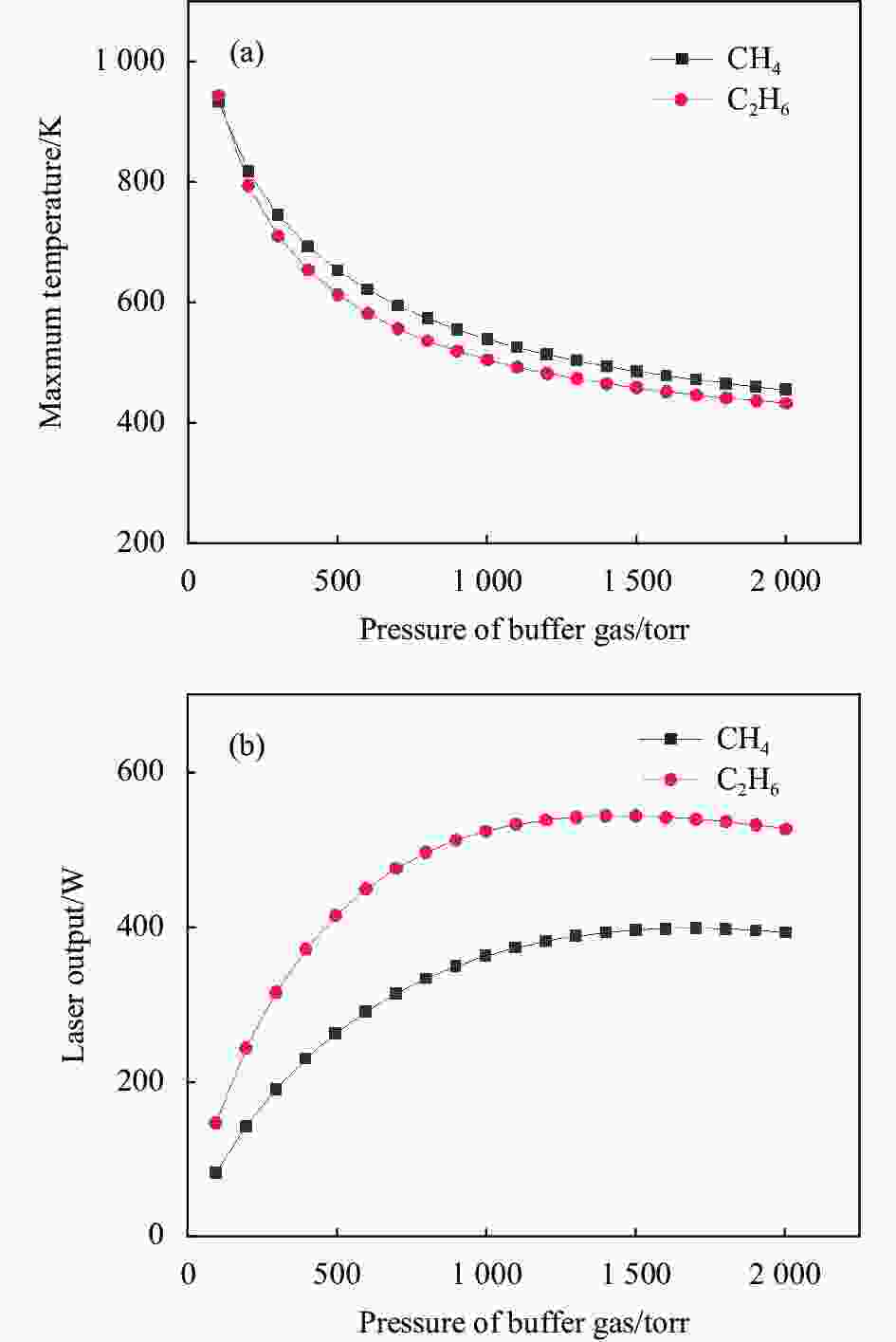
图4给出了使用纯CH4和纯C2H6作为缓冲气体时,在1 kW泵浦下,蒸汽池内气体最高温度和输出激光功率随缓冲气体的压强变化图。可以看出,当气体压强低于1500 torr时,气体温度越低,对应激光的输出功率越高,在同一压强下,相比于CH4,C2H6作为缓冲气体,激光输出功率要更高。在C2H6和CH4的压强超过1000 torr之后,精细结构能级自旋轨道弛豫逐渐趋向饱和,气体温度降低的速度放慢,激光输出功率的增速也减缓,当CH4和C2H6的压强为1700 torr和1500 torr时,对应最高激光输出功率分别为397.4 W和542.1 W;当气体压强继续升高时,激光输出功率不再受自旋轨道弛豫速率影响,而缓冲气体的相关热系数(摩尔热容、热导率、粘滞系数)受压强的影响较小,蒸汽池内温度趋向稳定,激光输出功率甚至出现下降,因此,烷烃类气体压强不能过高。
-
图5所示为1 kW泵浦下使用烷烃(CH4或C2H6)和惰性气体(He或Ar)的混合气体作为缓冲气体,蒸汽池内温度三维分布与缓冲气体压强之间的关系,其中烷烃的压强保持在700 torr,惰性气体的压强分别为400 torr、700 torr。当混合气体中C2H6的压强为700 torr时,400 torr He和Ar对应蒸汽池内最高温度分别为515.7 K和520.7 K,700 torr He和Ar对应蒸汽池内最高温度分别为492.1 K和498.7 K;当混合气体中CH4的压强为700 torr时,400 torr He和Ar对应蒸汽池内最高温度分别为533.6 K和539.4 K,700 torr He和Ar对应蒸汽池内最高温度分别为501.2 K和508.1 K。可见,在同一烷烃的作用下,相同压强的He和Ar对应蒸汽池内温度非常接近,这是因为He和Ar的碰撞展宽系数和摩尔热容等物理特性非常接近。而在惰性气体的成分和压强相同时,CH4对应的蒸汽池内最高温度要高于C2H6。

图 5 使用混合气体作为缓冲气体时,蒸汽池内温度分布与缓冲气体压强之间的关系。(a) CH4 700 torr, He 400 torr; (b) CH4 700 torr, Ar 400 torr; (c) CH4 700 torr, He 700 torr; (d) CH4 700 torr, Ar 700 torr; (e) C2H6 700 torr, He 400 torr; (f) C2H6 700 torr, Ar 400 torr; (g) C2H6 700 torr, He 700 torr; (h) C2H6 700 torr, Ar 700 torr
Figure 5. Relationship between temperature distribution and buffer gas pressure in vapor cell using mixed gas as buffer gas. (a) 700 torr methane and 400 torr helium; (b) 700 torr methane and 400 torr argon; (c) 700 torr methane and 700 torr helium; (d) 700 torr methane and 700 torr argon;(e) 700 torr ethane and 400 torr helium; (f) 700 torr ethane and 400 torr argon; (g) 700 torr ethane and 700 torr helium; (g) 700 torr ethane and 700 torr argon
图6为6种缓冲气体对应蒸汽池内最高温度随气体总压强变化的曲线,其中图(a)和(b)中烷烃气体的压强分别为400 torr和700 torr。可见,对于这6种缓冲气体,蒸汽池内温度都随压强的增加不断降低,但是降低的速度逐渐放缓,温度逐渐趋向稳定。对于烷烃气体和He组成的混合气体,当气体总压强固定为1200 torr时,400 torr C2H6和CH4对应的蒸汽池内最高温度分别为514.8 K和521.0 K,700 torr C2H6和CH4对应的蒸汽池内最高温度分别为507.2 K和521.6 K,对比可知,在总压强相同时,混合气体中C2H6压强越高,蒸汽池内温度就越低,而CH4对蒸汽池内温度的降低效果则与惰性气体相差不大。因此,相比于其他缓冲气体,使用纯C2H6作为缓冲气体对蒸汽池内温度降低效果最明显。

图 6 不同缓冲气体对应蒸汽池内最高温度随气体总压强变化曲线。(a) 混合气体中烷烃压强为400 torr;(b) 混合气体中烷烃压强为700 torr
Figure 6. Curves of maximum temperature versus total gas pressure in vapor cell of various buffer gas. (a) 400 torr alkanes in mixed gases;(b) 700 torr alkanes in mixed gases
图7中的曲线分别表示在烷烃(CH4和C2H6)压强为200 torr、400 torr和700 torr时,输出激光功率随He或Ar的压强变化关系。在混合气体中,C2H6的压强为700 torr时,500 torr的He和Ar对应的激光输出分别为460.3 W和467.6 W;CH4压强为700 torr时,500 torr的He和Ar对应的激光输出分别为313.5 W和318.9 W。对比He和Ar对激光输出功率的影响不难发现,由于二者具有非常相近的物理性质,相同情况下对应蒸汽池内温度也十分接近,因此输出激光功率也十分接近。在混合气体中CH4压强为200 torr时,100 torr和500 torr He对应激光输出功率分别为150.3 W和166.3 W;在混合气体中C2H6压强为200 torr时,100 torr和500 torr He对应激光输出功率分别为250.8 W和268.4 W。可见,在烷烃压强较低时,Cs原子精细结构弛豫速率较低,适当增加惰性气体的压强能够少量提升弛豫速率,从而提升激光输出功率。在混合气体中CH4压强为700 torr时,100 torr和500 torr He对应激光输出功率分别为314.7 W和313.5 W;在混合气体中C2H6压强为700 torr时,100 torr和500 torr He对应激光输出功率分别为472.6 W和460.3 W。可见,随着烷烃压强的增加,Cs原子精细结构弛豫逐渐提升,增加惰性气体的压强会导致Cs原子受激吸收和发射截面减小,这是激光输出功率下降的主要因素。
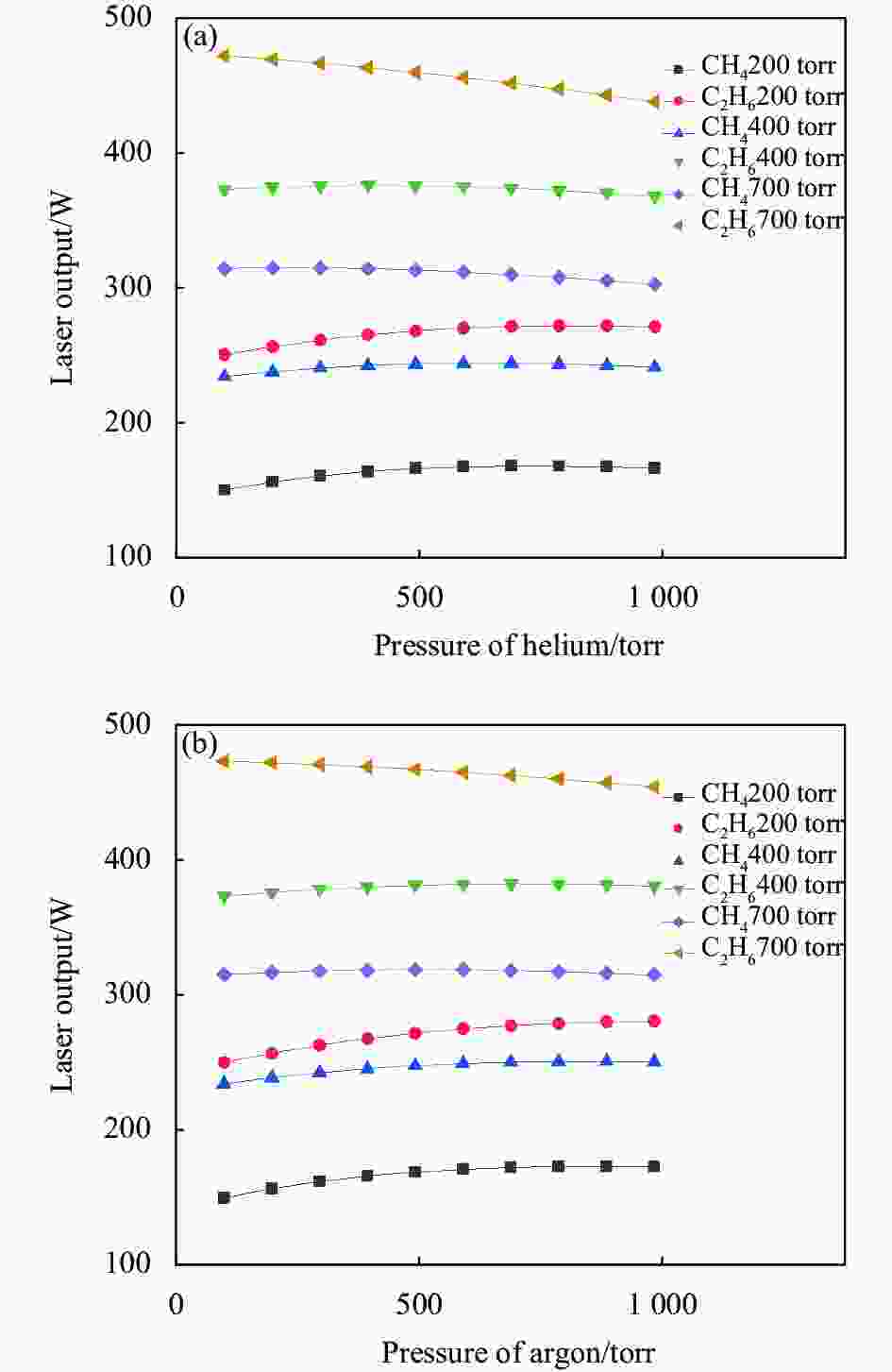
图 7 不同缓冲气体对应激光输出功率随He (a)和Ar (b)的压强变化曲线
Figure 7. Curves of laser output power versus pressure of helium (a) and argon (b) of various buffer gases
将图7与图4对比可知,在总压强相同时,使用纯C2H6作为缓冲气体时蒸汽池内温度较低,并且激光输出功率较高。因此,如果不考虑高温下的化学反应,相同条件下相比于其他缓冲气体,纯C2H6是最合适的缓冲气体。
-
文中在速率方程、流体力学方程与热平衡方程的基础上,首次将光束传播方程引入循环流动Cs-DPAL理论模型,分析了高功率泵浦的情况下,缓冲气体成分和压强对循环流动Cs-DPAL蒸汽池内气体温度三维分布和输出特性的影响。仿真结果表明,使用纯烷烃气体作为缓冲气体,C2H6对应的蒸汽池内温度要明显低于CH4,输出功率要明显高于CH4,因此,相比于CH4,C2H6将是一个更好的选择。使用烷烃气体和惰性气体的混合物作为缓冲气体,相同条件下,He和Ar对应的激光输出和蒸汽池内温度均十分接近,烷烃的压强应该保持在较低水平,此时加入适量的He或Ar可降低蒸汽池内温度并提升激光输出功率。在总压强相同的条件下,相比于使用混合气体或纯CH4作为缓冲气体,使用纯C2H6作为缓冲气体可获得最低的蒸汽池内温度分布和最高的激光输出。
Effect of buffer gas on gas temperature distribution and output characteristics of flowing-gas circulation cesium vapor laser
-
摘要: 为研究缓冲气体对循环流动半导体泵浦碱金属蒸汽激光器(DPAL)气体温度三维分布和输出性能的影响,将光束传播方程引入循环流动DPAL理论模型,仿真分析了高功率泵浦情况下缓冲气体配比和压强对端面泵浦横向流动铯蒸汽激光器(Cs-DPAL)输出性能的影响,获得了蒸汽池内工作气体温度的三维分布和对应的输出功率。结果表明,使用纯烷烃作为缓冲气体时,相比于CH4,相同压强的C2H6对应的蒸汽池内温度更低,激光输出功率更高;使用烷烃气体和惰性气体的混合物作为缓冲气体时,若烷烃的压强较低,加入适量的He或Ar可降低蒸汽池内温度并提升激光输出功率。Abstract: To study the effect of buffer gas on the three-dimensional gas temperature distribution and output performance of flowing-gas circulation diode-pumped alkali vapor laser (DPAL), the beam propagation equation is introduced into the theoretical model of flowing-gas circulation DPAL in this paper. The effects of the composition and pressure of the buffer gas on the output performance of the end-pumped transverse flow cesium vapor laser are simulated and analyzed. The three-dimensional distribution of work temperature and output power are obtained. The results show that when using pure alkanes as buffer gas, the temperature in the vapor cell corresponding to ethane is lower than methane with same pressure, and the laser output power is higher. When the mixture of alkane gas and inert gas is used as a buffer gas, if the pressure of alkane is low, adding an appropriate amount of He or Ar can reduce the temperature in the vapor cell and increase the laser output power.
-
Key words:
- DPAL /
- flowing-gas circulation /
- high power laser
-
图 3 蒸汽池内温度分布与缓冲气体压强之间的关系。(a)、(c)、(e)对应CH4压强分别为400 torr、700 torr和1000 torr;(b)、(d)、(f)对应C2H6压强分别为400 torr、700 torr和1000 torr
Figure 3. Relationship between temperature distribution and buffer gas pressure in vapor cell. (a), (c) and (e) correspond to 400 torr, 700 torr and 1000 torr methane pressures, respectively; (b), (d) and (f) correspond to 400 torr, 700 torr and 1000 torr ethane pressures, respectively
5 使用混合气体作为缓冲气体时,蒸汽池内温度分布与缓冲气体压强之间的关系。(a) CH4 700 torr, He 400 torr; (b) CH4 700 torr, Ar 400 torr; (c) CH4 700 torr, He 700 torr; (d) CH4 700 torr, Ar 700 torr; (e) C2H6 700 torr, He 400 torr; (f) C2H6 700 torr, Ar 400 torr; (g) C2H6 700 torr, He 700 torr; (h) C2H6 700 torr, Ar 700 torr
5. Relationship between temperature distribution and buffer gas pressure in vapor cell using mixed gas as buffer gas. (a) 700 torr methane and 400 torr helium; (b) 700 torr methane and 400 torr argon; (c) 700 torr methane and 700 torr helium; (d) 700 torr methane and 700 torr argon;(e) 700 torr ethane and 400 torr helium; (f) 700 torr ethane and 400 torr argon; (g) 700 torr ethane and 700 torr helium; (g) 700 torr ethane and 700 torr argon
表 1 循环流动Cs-DPAL仿真参数
Table 1. Parameters of gas flowing diode pumped cesium laser
Parameters Value Parameters Value Central wavelength of pump light/nm 852.3 Waist radius of pump light/mm 1 Pump light linewidth/GHz 70 Pump light transmittance of M1 and M2 100% Pump light transmittance of M3 99% Laser reflectivity of M4 50% Distance between waist of pump light and central of vapor cell/mm 0 Sidewall temperature of vapor cell/℃ 100 Distance between vapor cell and M3/mm 50 Distance between vapor cell and M4/mm 50 Gain length of vapor cell/mm 20 Velocity of flowing gas/m·s−1 20 表 2 缓冲气体的摩尔热容、热导率和粘滞系数
Table 2. Molar heat capacity, thermal conductivity and viscosity of buffer gases
Buffer gases Molar heat capacity/J·mol−1·K−1 Thermal conductivity/ W·m−1·K−1 Viscosity/ Pa·s−1 Ethane 72.027 4.464×10−2 1.353×10−5 Methane 43.550 5.906×10−2 1.566×10−5 Helium 20.785 2.066×10−1 2.636×10−5 Argon 20.810 2.451×10−2 3.138×10−5 -
[1] Ji Yanhui, He Yang, Wan Haohua, et al. Research progress on the high power flowing-gas circulation diode-pumped alkali vapor laser (Invited) [J]. Infrared and Laser Engineering, 2020, 49(12): 20201080. (in Chinese) doi: 10.3788/IRLA20201080 [2] Zhdanov B V, Rotondaro M D, Shaffer M K, et al. Potassium diode pumped alkali laser demonstration using a closed cycle flowing system [J]. Optics Communications, 2015, 354: 256-258. doi: 10.1016/j.optcom.2015.06.010 [3] Zhdanov B V, Rotondaro M D, Shaffer M K, et al. Low-pressure cesium and potassium diode pumped alkali lasers: pros and cons [J]. Optical Engineering, 2016, 55(2): 026105. doi: 10.1117/1.OE.55.2.026105 [4] Gavrielides A, Schlie L A V, Loper R D, et al. Analytic treatment of beam quality and power efficiency in a high-power transverse flow diode pumped alkali laser [J]. JOSA B, 2018, 35(9): 2202-2210. doi: 10.1364/JOSAB.35.002202 [5] Bogachev A V, Garanin S G, Dudov A M, et al. Diode-pumped caesium vapour laser with closed-cycle laser-active medium circulation [J]. Quantum Electronics, 2012, 42(2): 95-98. doi: 10.1070/QE2012v042n02ABEH014734 [6] Knize R J, Zhdanov B V, Rotondaro M D, et al. Experimental study of the Cs diode pumped alkali laser operation with different buffer gases [J]. Optical Engineering, 2016, 55(3): 036109. doi: 10.1117/1.OE.55.3.036109 [7] Yamamoto T, Yamamoto F, Endo M, et al. Experimental investigation of gas flow type DPAL[C]//XXI International Symposium on High Power Laser Systems and Applications 2016. International Society for Optics and Photonics, 2017, 10254: 102540S. [8] Yacoby E, Auslender I, Barmashnko B D, et al. Continuous wave diode pumped flowing-gas cesium laser[C]//XXII Inter-national Symposium on High Power Laser Systems and Applications. International Society for Optics and Photonics, 2019, 11042: 110420D. [9] Xu Yan, Chen Fei, Xie Jijiang, et al. Influence of buffer gas on performance of alkali vapor laser [J]. Infrared and Laser Engineering, 2015, 44(2): 455-460. (in Chinese) doi: 10.3969/j.issn.1007-2276.2015.02.010 [10] Shu H, Bass M. Three-dimensional computer model for simu-lating realistic solid-state lasers [J]. Applied Optics, 2007, 46(23): 5687-5697. doi: 10.1364/AO.46.005687 [11] Han J, Wang Y, Cai H, et al. Algorithm for evaluation of temperature distribution of a vapor cell in a diode-pumped alkali laser system (part II) [J]. Optics Express, 2015, 23(7): 9508-9515. doi: 10.1364/OE.23.009508 [12] Shen B, Pan B, Jiao J, et al. Kinetic and fluid dynamic modeling, numerical approaches of flowing-gas diode-pumped alkali vapor amplifiers [J]. Optics Express, 2015, 23(15): 19500-19511. doi: 10.1364/OE.23.019500 [13] Waichman K, Barmashenko B D, Rosenwaks S. CFD DPAL modeling for various schemes of flow configurations[C]//Proceedings of SPIE, 2014, 9251: 92510U. [14] Friend D G, Ingham H, Fly J F. Thermophysical properties of ethane [J]. Journal of physical and Chemical Reference Data, 1991, 20(2): 275-347. doi: 10.1063/1.555881 [15] Friend D G, Ely J F, Ingham H. Thermophysical properties of methane [J]. Journal of Physical and Chemical Reference Data, 1989, 18(2): 583-638. doi: 10.1063/1.555828 [16] Span R, Lemmon E W, Jacobsen R T, et al. A reference equation of state for the thermodynamic properties of nitrogen for temperatures from 63.151 to 1000 K and pressures to 2200 MPa [J]. Journal of Physical and Chemical Reference Data, 2000, 29(6): 1361-1433. doi: 10.1063/1.1349047 [17] Arp V D, McCarty R D. Thermophysical properties of Helium-4 from 0.8 to 1500 K with pressures to 2000 MPa[R]. Boulder, CO, USA: National Institute of Standards and Technology, 1989. [18] Stewart R B, Jacobsen R T. Thermodynamic properties of argon from the triple point to 1200 K with pressures to 1000 MPa [J]. Journal of Physical and Chemical Reference Data, 1989, 18(2): 639-798. doi: 10.1063/1.555829 -






 下载:
下载: