-
2,4,6,8,10,12-六硝基-2,4,6,8,10,12-六氮杂异伍兹烷,简称HNIW或CL-20,是目前已知的密度最大、能量最强的非核单质炸药[1-3],在国防军事领域具有广阔的应用前景。CL-20在常温常压下存在α、β、γ和ε四种晶相,其中ε-CL-20能量密度最高、热稳定性最好,具有最高的实际应用价值。研究表明,在特定的温度压力等外界载荷作用下,上述晶相会发生相互转化[4]。这种相转换通常会导致晶体的体积膨胀和应力开裂,由此产生的缺陷可能会成为炸药潜在剪切带和热点的来源[5-6],引起武器弹药的变形、破坏和失效。因此,研究炸药的相变机理,对满足现代武器装备服役安全性和可靠性要求具有重要意义。
为了深入理解CL-20相变的敏感机制,国内外研究人员采用多种技术手段开展了广泛研究。相较于传统的热重分析(TGA)等方法而言,光学手段具有非接触、多分子尺度动态表征等优点,是炸药相变研究的常用技术方法。通过X射线衍射技术(XRD)对CL-20热诱导相变的研究[7]从分子结构和晶格参数方面表征了演化特性。相变前后硝基取向及晶格参数不同,晶胞体积膨胀,晶体形貌出现裂纹;利用红外光谱(IR)技术[8]对热加载CL-20红外吸收的研究表明,分子各基团温度响应特性不同,骨架外-NO2、C-H和骨架内C-N振动经历了热膨胀、热致相变、热分解三个阶段,骨架内C-C振动出现与相变密切相关的新特征峰;采用拉曼光谱技术(Raman)对CL-20相变的研究主要集中在压力诱导方面,通过拉曼峰演变解析发现[9],N-N伸缩振动拉曼峰的合并、C-N-N弯曲振动峰的合并和增强以及C-H弯曲振动峰的分裂是新相产生的显著特征。上述研究表明,CL-20相变的微观机制与分子结构、基团及化学键振动等密切相关。但有报道表明[10],晶体的分子间相互作用是炸药多晶型形成和转换的主要作用力。因此,开展CL-20相变过程分子间弱相互作用的研究对进一步理解其复杂物理机制具有重要意义。
太赫兹(THz)波是指频率位于0.1~10.0 THz (波长3 mm~30 μm)范围内的电磁辐射,其频率高、能量低的特性可实现无损检测[11-13],更重要的是该频段涵盖了晶体分子堆积方式和氢键网络等弱分子间作用引起的特定振动模式。因此,相比于红外和拉曼光谱,太赫兹光谱对晶体的晶型/构象更加敏感,有望成为炸药相变研究的新型技术手段。但是,太赫兹频段不存在特定振动模式的过渡,仅通过测量光谱难以进一步理解其光谱特性形成的物理机制。密度泛函理论(DFT)是一种研究多电子体系电子结构的常用方法,近年来利用DFT开展分子晶体的太赫兹频段振动模式分析已经获得广泛应用[14-15],可以对CL-20的太赫兹光谱研究提供必要的理论支持。
综上所述,为了深入理解CL-20相变物理机制,利用THz-TDS结合DFT计算开展温度加载太赫兹光谱特性研究。该研究对进一步理解CL-20在热刺激下发生相变乃至最终分解的复杂物理机制以及研制性能优异的新型CL-20基炸药具有重要意义。
-
CL-20晶体来自中国工程物理研究院化学材料研究所。制备工艺为:首先以苄胺和乙二醛为原料(摩尔比2.2∶1),在介质乙腈/水(体积比11∶1)中以有机酸为催化剂在0 ℃下缩合16~18 h,制取六苄基六氮杂异伍兹烷(HBIW)。然后以Pd/C作为催化剂,乙酸酐作为溶剂,溴苯作为氢解助剂,将HBIW在氢气中氢解18 h。再用热氯仿对产物进行萃取、蒸馏得到粗品,用乙腈进行重结晶得到较纯的四乙酰基二苄基六氮杂异伍兹烷(TADBIW)。最后对TADBIW二次脱苄并分离产物,再将脱苄产物硝化,制得CL-20。
实验采用的测量样品为直径13 mm、厚度1.5~2.0 mm的圆形药片。制备流程为:取适量CL-20晶体在玛瑙研钵中轻轻研磨,通过200目筛过滤得到颗粒直径小于74 μm的样品粉末。再取70 mg样品粉末与400 mg聚四氟乙烯(PTFE)混合均匀,经压片机压制成药片。PTFE是一种熔点高(327 ℃)且对太赫兹波段吸收弱的合成材料,在样品制备中加入PTFE既能有效防止CL-20过度吸收,还可大幅提升样品的压制成型性能。
-
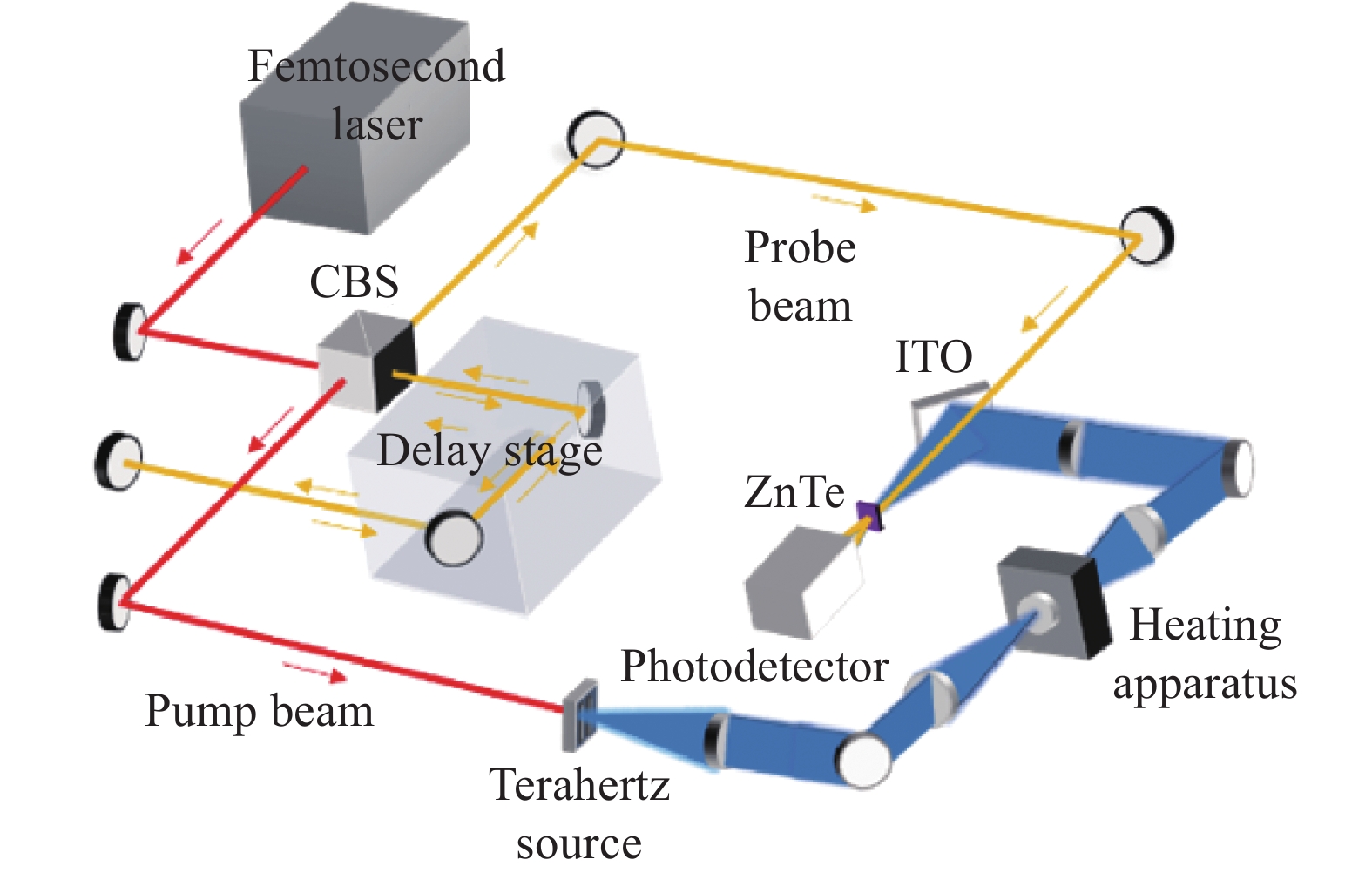
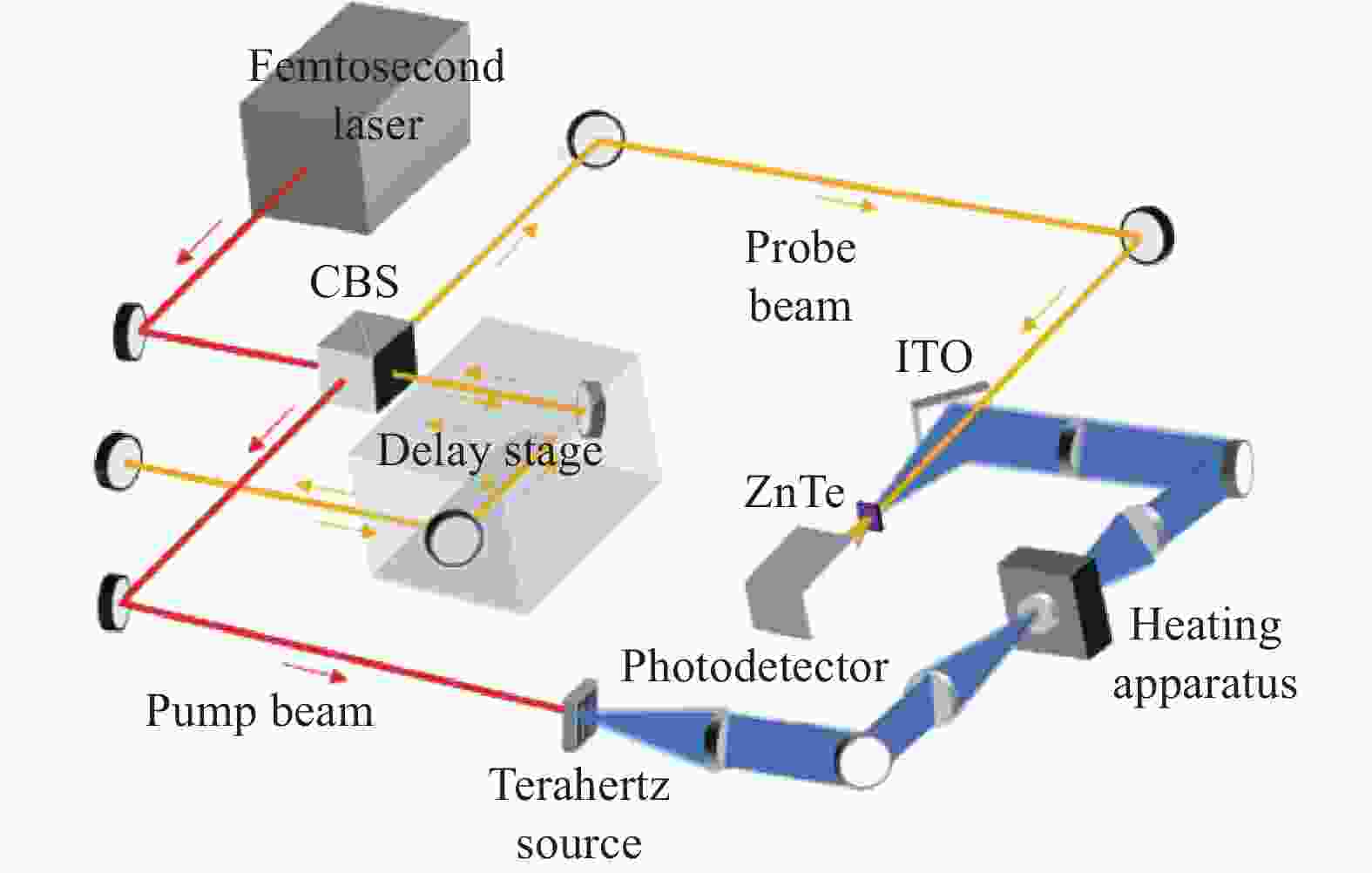
实验采用的太赫兹时域光谱系统(THz-TDS)主要由飞秒激光器(MaiTai,Spectra-Physics)和太赫兹时域光谱测试仪(Z-3,Zomega)构成,如图1所示。系统的工作原理为:首先,由飞秒激光器产生中心频率为800 nm,脉宽为80 fs,震荡频率为80 MHz的激光脉冲,并经过偏振分束棱镜(CBS)分离为100 mW和20 mW的泵浦光与探测光。泵浦光经反射和透射镜组聚焦入射至光导天线间隙(200 μm),产生光生载流子,根据麦克斯韦方程[16],光生载流子在外加电场的调制下辐射出太赫兹波。探测光在经过延迟平移台后与携带有样品信息的太赫兹波共线入射至碲化锌<110>晶体(ZnTe)(厚度2 mm),由光电探测器实现相干探测。为避免水分引起的太赫兹波强吸收对实验产生干扰,将系统置于密闭空间并持续充入干燥空气。测量环境条件保持相对湿度<1%,温度23 ℃。
-
将THz-TDS系统测量的参考信号和样品信号分别记为
$ {E_{{\text{ref}}}}\left( t \right) $ 和$ {E_{{\text{sam}}}}\left( t \right) $ ${{E}}_{\mathrm{s}\mathrm{a}\mathrm{m}}\left({t}\right)$ 。其中,参考信号为太赫兹波经过干燥空气的信号,样品信号为太赫兹波透过待测样品的信号。对上述时域信号分别进行快速傅里叶变换,得到频域信号$ {E_{{\text{ref}}}}\left( \omega \right) $ 和$ {E_{{\text{sam}}}}\left( \omega \right) $ 。通过公式(1)和(2)计算获得振幅比$ \rho \left( \omega \right) $ 和相位差$ \varphi \left( \omega \right) $ :$$ \rho \left( \omega \right) = \frac{{\mathop A\nolimits_{{\text{sam}}} }}{{\mathop A\nolimits_{{\text{ref}}} }} $$ (1) $$ \varphi \left( \omega \right) = \mathop \varphi \nolimits_{{\text{sam}}} - \mathop \varphi \nolimits_{{\text{ref}}} $$ (2) 折射率
${n}(\omega)$ 和吸收系数${α}(\omega)$ 计算公式如下所示:$$ n\left( \omega \right) = \varphi \left( \omega \right)\frac{c}{{\omega d}} + 1 $$ (3) $$ \alpha \left( \omega \right) = \frac{2}{d}\ln \left( {\frac{{4n\left( \omega \right)}}{{\rho \left( \omega \right){{\left[ {n\left( \omega \right) + 1} \right]}^2}}}} \right) $$ (4) 式中:c为空气中光速;ω为角频率(
$ \omega = 2\pi f $ );d为样品厚度。计算获得的吸收数据通过Origin2018软件中的Savitzky-Golay方法进行平滑处理,窗口点数设置为33,多项式阶数为5。基于DFT对ε和γ相CL-20的低频振动特性开展模拟计算。DFT计算利用Materials Studio软件包的DMol3模块来完成,使用的晶胞参数来自于剑桥晶体数据库(Cambridge Crystallographic Data Centre, CCDC)。计算过程中首先根据能量最小化原则对CL-20进行几何结构优化,在优化结果偏差较小的基础上开展振动频率和强度计算,计算结果最终通过洛伦兹曲线拟合绘制成吸收谱。
-
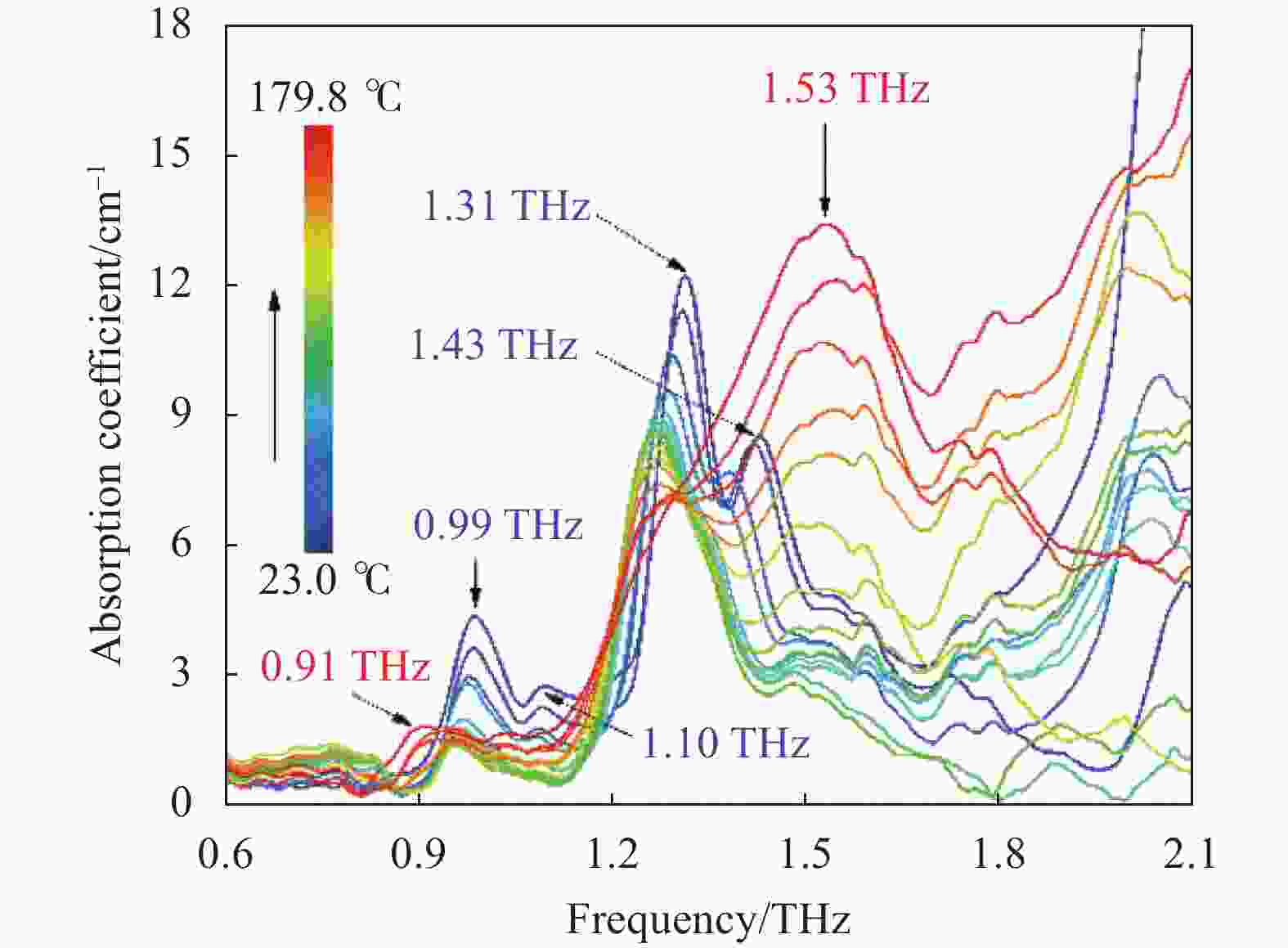
图2描述了在23.0~179.8 ℃范围内,CL-20太赫兹吸收光谱随温度的演化过程。常温下CL-20存在四处明显吸收,分别位于0.99、1.10、1.31、1.43 THz。在23~111 ℃温度区间中,四处吸收均出现中心位置红移和强度降低。这是由于温度的升高导致CL-20晶体膨胀,削弱了分子间作用力。同时可以看出,不同吸收的频移和强度衰减程度存在差异,如:0.99、1.31 THz吸收峰红移至0.96 THz和1.27 THz,频率偏移Δf为0.03 THz和0.04 THz,而1.10、1.43 THz吸收峰逐渐消失,这说明引起上述吸收的分子间作用力对热刺激的响应机制并不相同。在111.0~132.5 ℃温度范围内各吸收变化微弱,处于相对稳定阶段,晶体热膨胀作用造成的影响达到极限。当温度达到136.8 ℃时,1.4~1.6 THz频率区间的吸收强度开始逐渐增加。随着温度持续升高,吸收进一步增强,最终以1.53 THz为中心形成一个新的宽频吸收峰。与此同时,1.31 THz吸收逐渐削弱至消失,而0.99 THz吸收继续红移至0.91 THz,强度有所增加。图3描绘了1.53 THz频率点吸收强度随温度升高的演变趋势,显而易见的,强度从136.8 ℃开始迅速增加,曲线陡然升高,意味着出现了新的分子振动模式。通常,随着温度的上升,吸收峰呈频率红移和强度衰减趋势,而1.53 THz吸收异常的快速增强现象表明在136.8 ℃开始发生相变。

图 3 升温过程中1.53 THz吸收强度变化
Figure 3. Variation of absorption intensity at 1.53 THz frequency position during heating process
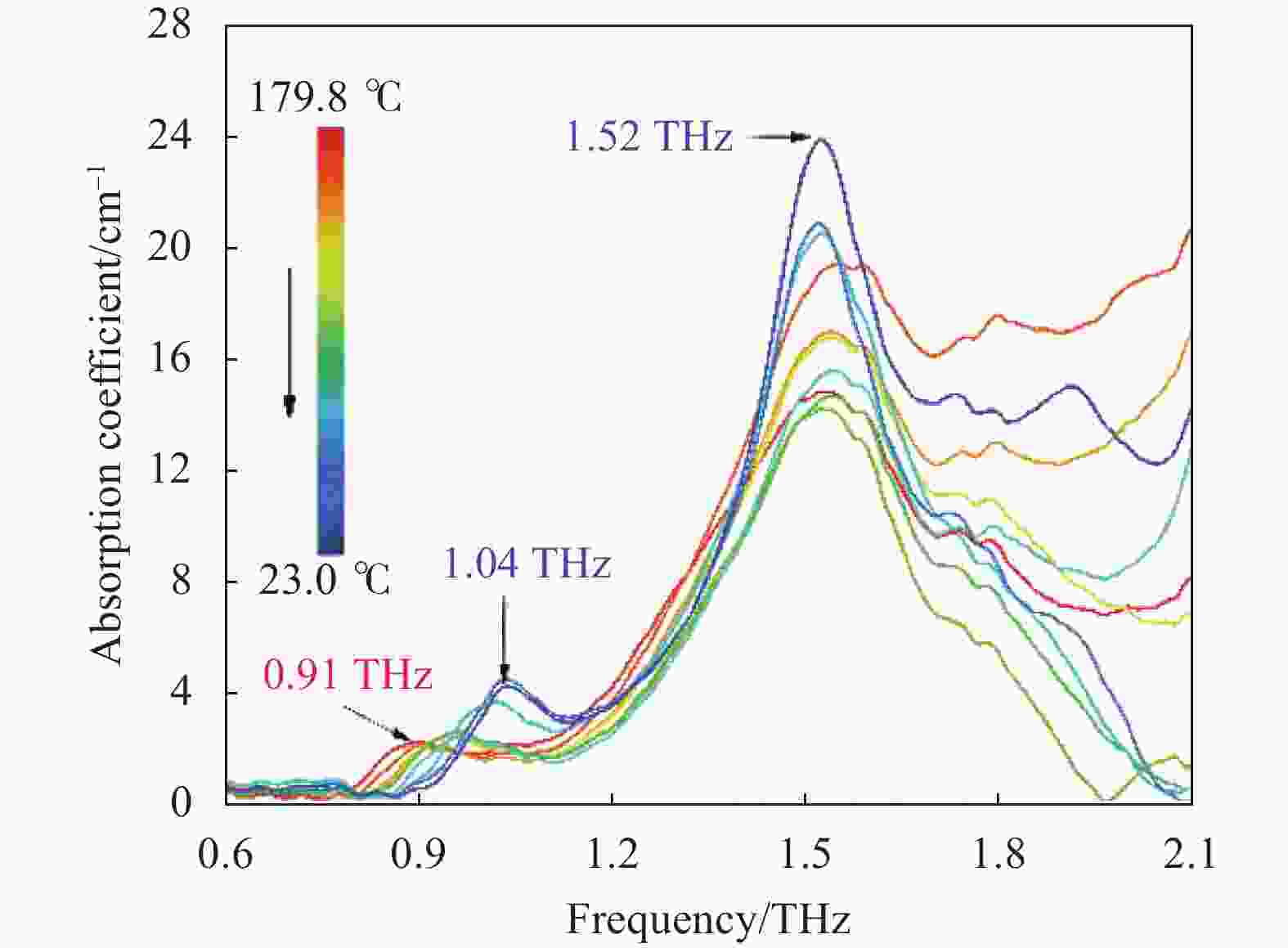
除了升温过程以外,还获取了CL-20自然冷却至室温的吸收谱,如图4所示。冷却后的光谱显著区别于加热前,这表明升温中发生的相变是不可逆的。0.91 THz吸收在降温中出现明显的蓝移和增强现象,吸收峰中心最终稳定在1.04 THz。蓝移的原因是温度的降低使晶体单元体积收缩,振动的有效力常数增加,导致其以更高频率振动。此外,与相变相关的新宽频吸收在冷却中呈增强和尖锐化趋势,这是由于晶体体积减小使分子间作用力增强导致。并且,该吸收在冷却中的强度波动反映了晶体的不稳定特征。
为了进一步分析实验光谱,依据DFT对ε和γ相CL-20晶体低频振动进行计算,计算结果与实验光谱的对比如图5所示,红色和黑色实线分别表示实验吸收光谱和DFT计算结果,黑色虚线描述了计算获得的每个振动模式。将23 ℃实验光谱中的0.99、1.10、1.31 THz吸收分别归结于ε-CL-20计算的0.96、1.16、1.30 THz振动模态,频率偏差为0.03、0.06、0.01 THz,吸收强度也存在一定差异,1.43 THz吸收未在计算中体现。将179.8 ℃光谱中0.91 THz和1.53 THz吸收归结于γ-CL-20计算的0.78 THz和1.53 THz振动模态,频率偏差为0.13 THz和0 THz,0.91 THz吸收峰的强度估计较高。对光谱中各吸收的指认证明在136.8~179.8 ℃太赫兹光谱中观测到的不可逆转变为ε→γ-CL-20相变。
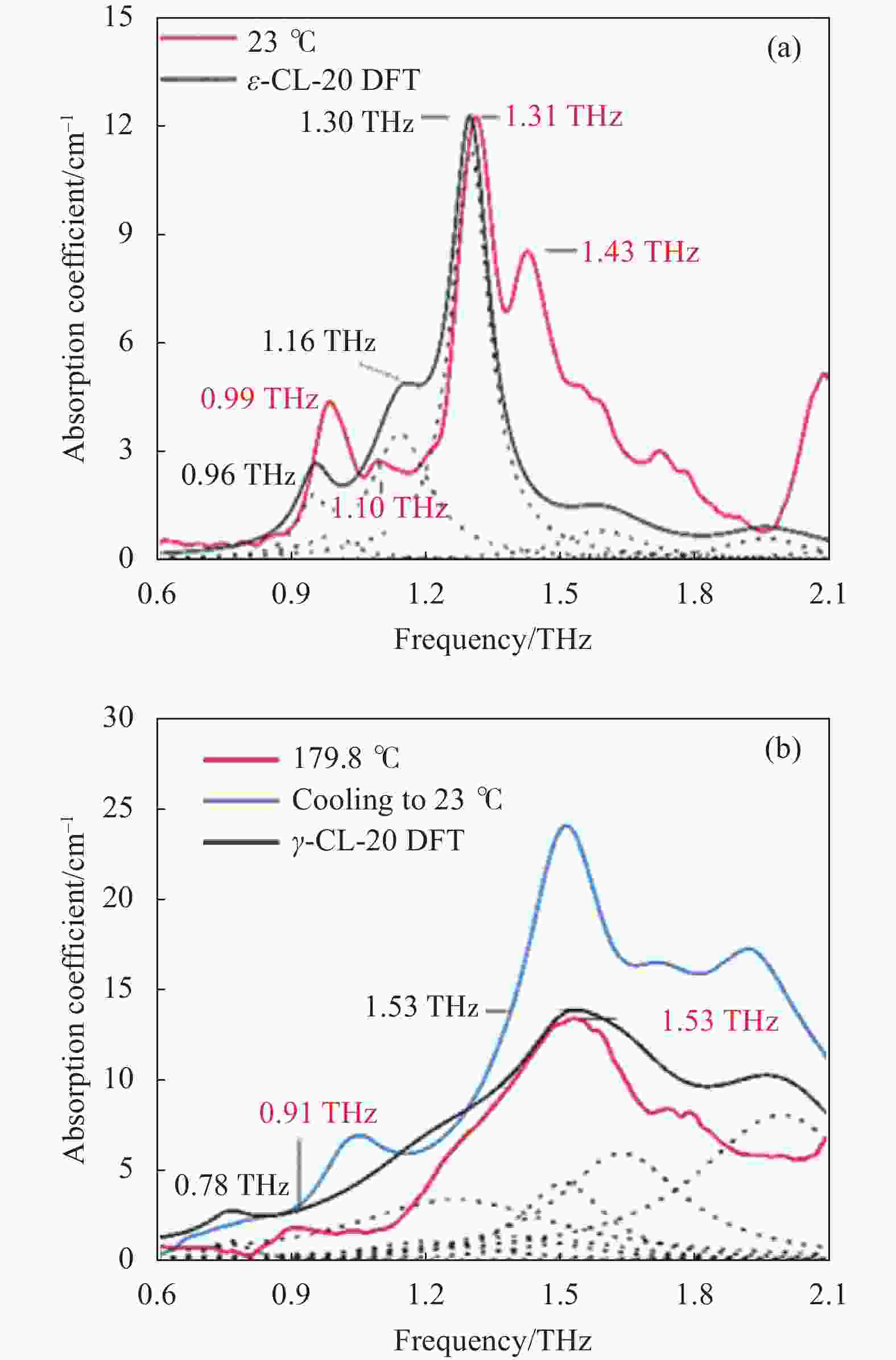
图 5 DFT计算结果与实验光谱。 (a) ε-CL-20;(b) γ-CL-20
Figure 5. DFT calculation results and experimental spectra. (a) ε-CL-20; (b) γ-CL-20
图6描绘了ε相CL-20各个吸收的振动模态,图中红、蓝、灰、白色小球分别代表氧、氮、碳、氢原子,绿色箭头表示原子振动矢量。0.99 THz和1.10 THz吸收均源于笼型骨架外围硝基基团的旋转振动,前者围绕b轴旋转,后者围绕分子笼心且与c轴和∠aob角平分线构成的平面平行。0.99 THz振动模态中N2、N4原子所在硝基振动矢量最强,而1.10 THz振动由N2、N4、N10所在硝基主导,这些硝基参与的分子内和分子间氢键作用在CL-20中占比较小,这是导致两个吸收峰较弱的原因。并且,二者振动方向的差异造成了不同的温度响应特性。1.31 THz吸收由分子在c轴方向的平移振动引起,笼型骨架明显参与了该模态,分子间占主导的范德华相互作用是振动的主要来源。这种平移振动使c轴方向上热膨胀系数更大,导致1.31 THz吸收红移程度更强。该吸收在升温中逐渐消失,表明CL-20笼型骨架的振动模态发生了改变,这是相变发生的内在原因。

图 6 ε-CL-20各吸收峰振动模态。(a) 0.99 THz;(b) 1.10 THz;(c) 1.31 THz
Figure 6. Vibration modes of ε-CL-20 absorption peaks. (a) 0.99 THz; (b) 1.10 THz; (c) 1.31 THz
γ-CL-20的分子振动模态如图7所示。0.91 THz吸收由分子外围硝基基团围绕c轴的旋转振动引起,N4、N6、N10、N12原子所在硝基是振动贡献的主体,这种旋转源于晶体中的氢键网络相互作用。此外,0.91 THz吸收是由常温0.99 THz演变而来,其振动模态由围绕b轴向围绕c轴转变,这种演化与分子堆积方式转变密切相关。1.53 THz吸收源于分子的不平衡扭转振动,扭转方向沿反c轴偏向a轴,硝基与笼型骨架的共同振动导致了1.53 THz的较强吸收。ε→γ-CL-20相变前后,笼型骨架在1.31 THz沿c轴的平移振动消失,形成了1.53 THz的不平衡扭转振动模态。通常,CL-20分子骨架外围硝基参与氢键作用网络的构成,而上述笼型骨架振动模态的显著变化与分子间广泛的范德华力密切相关。此外,N2、N12所在硝基基团及相邻C-C、C-H键振动矢量明显更强,这是相变过程中N8原子所在硝基的方向扭转使分子对称性显著下降导致。
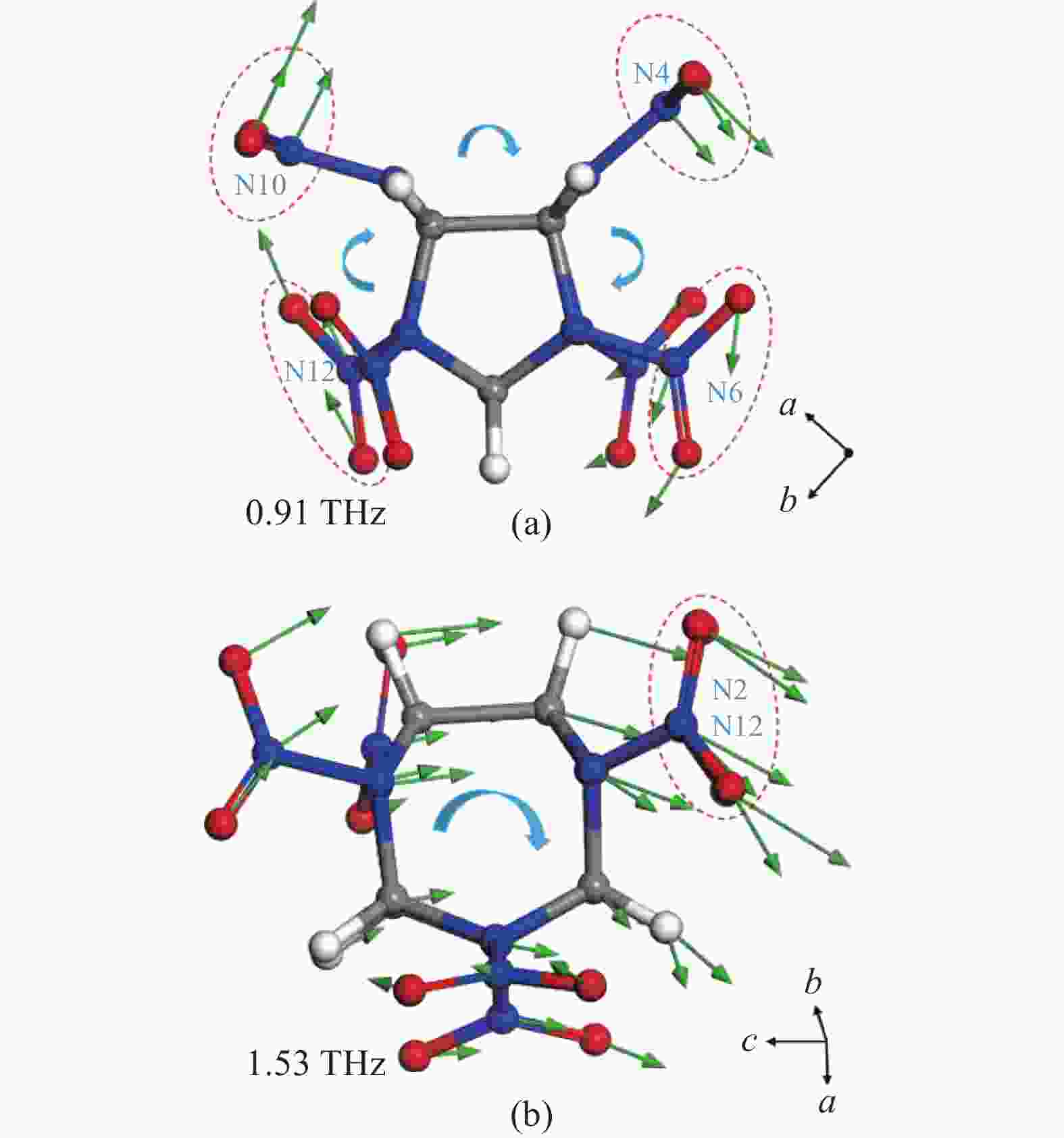
图 7 γ-CL-20各吸收峰振动模态。 (a) 0.91 THz;(b) 1.53 THz
Figure 7. Vibration modes of γ-CL-20 absorption peaks. (a) 0.91 THz; (b) 1.53 THz
在热刺激的作用下,CL-20分子笼型骨架和骨架外硝基均发生了振动模态变化。其中,笼型骨架振动模态的改变是ε→γ-CL-20相变的一个最显著的特征,这直接导致了常温1.31 THz吸收的消失以及与γ晶型相关的1.53 THz吸收的产生。此外,相较于笼型骨架,分子外围硝基对温度响应阈值更低,更易摆动。相变中,硝基发生方向扭转,氢键网络重构,晶体堆积方式改变,分子旋转振动模态因此发生变化。上述研究表明,分子间的弱相互作用在CL-20相变中扮演了重要角色,利用太赫兹光谱开展分子间作用力研究对理解炸药相变演化机制具有重要意义。
-
利用THz-TDS对CL-20升温过程的低频吸收光谱进行了测量,结合DFT计算鉴别了以136.8 ℃为起始温度的ε→γ-CL-20相变。光谱的特征变化表现为0.99 THz吸收的持续迁移和1.10、1.31、1.43 THz吸收的消失以及1.53 THz新的宽频吸收的产生。低频振动分析表明,由分子间占主要地位的范德华力引起的笼型骨架振动模态发生了显著改变,这是与γ相有关的新吸收的产生原因。此外,分子骨架外围硝基的旋转振动模态演变来源于晶体中氢键网络的重构,而0.99 THz吸收的持续演化与堆积方式的改变相关。研究表明,THz-TDS提供了一种认识CL-20相变中分子间弱相互作用机理的独特视角,对深入理解炸药相变复杂物理机制,设计、合成新型优质炸药具有重要的指导意义。
Terahertz spectral properties of temperature induced phase transition of CL-20
-
摘要: 2,4,6,8,10,12-六硝基-2,4,6,8,10,12-六氮杂异伍兹烷(HNIW, CL-20)是一种具有笼形结构的高能化合物,在军事及民用领域具有广阔的应用前景。为了深入理解CL-20晶体相变物理机制,采用太赫兹时域光谱技术(THz-TDS)对23.0~179.8 ℃升温范围内的吸收光谱进行了研究。热作用下太赫兹光谱的显著变化表明136.8 ℃时CL-20开始发生不可逆相变,结合固态密度泛函理论(DFT)计算鉴别该转变为ε→γ相变。并且,对CL-20低频振动特性的分析表明,分子笼型骨架的振动模态在相变中发生了显著改变,分子间广泛的范德华相互作用是其重要来源。此外,骨架外硝基基团的旋转振动演变与分子的氢键作用密切相关。该研究为进一步理解CL-20在高温加载下发生相变乃至爆轰/爆燃过程中的复杂物理机制提供了参考,对新型CL-20基优质炸药的设计和合成具有重要意义。Abstract: 2,4,6,8,10,12-hexanitro-2,4,6,8,10,12-hexazazoisowoodane (HNIW, CL-20) is a high-energy com- pound with a cage structure, which has broad application prospect in military and civil fields. In order to further understand the physical mechanism of the phase transition of CL-20 crystal, the absorption spectra in the temperature range of 23.0-179.8 ℃ are studied by using the terahertz time-domain spectroscopy (THz-TDS). The significant change of terahertz spectrum under thermal action indicated that the irreversible phase transition of CL-20 begins at 136.8 ℃. Combined with the results of solid-state density functional theory (DFT), the transition is identified as ε→γ-CL-20. Moreover, the analysis of the low-frequency vibration characteristics of CL-20 shows that the vibration modes of the molecular cage skeleton change significantly during the phase transition. The extensive van der Waals interactions between molecules are the source of this change. In addition, the evolution of the rotational vibration of the nitro group outside the framework is closely related to the molecular hydrogen bonding. This study provides a reference for further understanding the complex physical mechanism of CL-20 phase transition and detonation/deflagration under temperature loading. It is of great significance for the design and synthesis of high-quality explosives based on CL-20.
-
-
[1] Gao D X, Huang J, Lin X H, et al. Phase transitions and chemical reactions of octahydro-1, 3, 5, 7-tetranitro-1, 3, 5, 7-tetrazocine under high pressure and high temperature [J]. RSC Advances, 2019, 9(10): 5825-5833. doi: 10.1039/C8RA10638J [2] Wei X F, Xu J J, Li H Z, et al. A comparative study of experiments and calculations on the polymorphisms of 2, 4, 6, 8, 10, 12-hexanitro-2, 4, 6, 8, 10, 12-hexaazaisowurtzitane (CL-20) precipitated by solvent/anti-solvent method [J]. Journal of Physical Chemistry C, 2016, 120(9): 5042-5051. doi: 10.1021/acs.jpcc.6b00304 [3] Molt R W, Bartlett R J, Watson T, et al. Conformers of CL-20 explosive and ab initio refinement using perturbation theory: Implications to detonation mechanisms. [J]. The Journal of Physical Chemistry A, 2012, 116(49): 12129-12135. doi: 10.1021/jp305443h [4] Ghosh M, Venkatesan V, Mandave S, et al. Probing crystal growth of ε-and α-CL-20 polymorphs via metastable phase transition using microscopy and vibrational spectroscopy [J]. Crystal Growth & Design, 2014, 14(10): 5053-5063. [5] Zhang Zhenyu, Huan Shi, Lu Fangyun, et al. Viscoplastic collapse mechanism of hot-spot formation in porous TNT explosives [J]. Energetic Materials, 1994, 2(2): 36-48. (in Chinese) [6] Yang R J, An H M, Tan H M. Combustion and thermal decomposition of HNIW and HTPB/HNIW propellants with additives [J]. Combustion and Flame, 2003, 135(4): 463-473. doi: 10.1016/j.combustflame.2003.08.008 [7] Liu Y, Li S, Wang Z, et al. Thermally induced polymorphic transformation of hexanitrohexaazaisowurtzitane (HNIW) investigated by in-situ X-ray powder diffraction [J]. Central European Journal of Energetic Materials, 2016, 13(4): 1023-1037. doi: 10.22211/cejem/62349 [8] Zhao Lang, Sun Jie, Sui Heliang, et al. Temperature response law of the internal and external groups of CL-20 molecule skeleton by in-situ infrared spectroscopy [J]. Chinese Journal of Energetic Materials, 2019, 27(10): 845-852. (in Chinese) [9] Sun X Y, Sui Z L, Wang J K, et al. Phase transition routes for epsilon- and gamma-CL-20 crystals under high pressures of up to 60 GPa [J]. Journal of Physical Chemistry C, 2020, 124(9): 5061-5068. doi: 10.1021/acs.jpcc.9b11224 [10] Bu R P, Xiong Y, Wei X F, et al. Hydrogen bonding in CHON-containing energetic crystals: A review [J]. Crystal Growth & Design, 2019, 19(10): 5981-5997. [11] Bi Lingzhi, Yuan Minghui, Zhu Yiming. Method of measuring steel bar in building by THz wave [J]. Infrared and Laser Engineering, 2019, 48(1): 0125003. (in Chinese) doi: 10.3788/IRLA201948.0125003 [12] Wu Xueyang, Yang Jingyi, Wang Yingying, et al. Detection of hydration number of tert-butyl alcohol solution based on terahertz technology [J]. Infrared and Laser Engineering, 2019, 48(1): 0125006. (in Chinese) doi: 10.3788/IRLA201948.0125006 [13] Wang Yuye, Chen Liyu, Xu Degang, et al. Advances in terahertz three-dimensional imaging techniques [J]. Chinese Optics, 2019, 12(1): 1-18. (in Chinese) doi: 10.3788/co.20191201.0001 [14] Huang F, Liu M M, Xie M Q, et al. A combined study on the skeletal vibration of aminopyrine by terahertz time-domain spectroscopy and DFT simulation [J]. Optik, 2020, 208: 163913. doi: 10.1016/j.ijleo.2019.163913 [15] Chen T, Li Z, Zhang H, et al. Terahertz spectroscopic investigation of D- and DL-tartaric acid [J]. Chemical Physics Letters, 2019, 731: 136579. doi: 10.1016/j.cplett.2019.07.007 [16] Liang J, Zhang X X, Wang N, et al. Vibrational spectra of pyrazinamide and isoniazid studied by terahertz spectroscopy and density functional theory [J]. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2019, 228: 117591. -






 下载:
下载: